It accounts for 12% of New Zealand's total greenhouse gas emissions. Most nitrous oxide emissions arise from agriculture. The Government has set a target of reducing nitrous oxide emissions to net zero by 2050, meaning on-farm action is critical.
Actions to reduce nitrous oxide
-
Stocking rate and performance
Modelling shows it might be possible to reduce total greenhouse gas emissions by up to 10% on some farms, by fine-tuning production systems so the same output is obtained from fewer animals.
-
Efficiency improvements
Increasing outputs relative to inputs won't necessarily reduce absolute emissions, but it will improve emissions per unit of product. It's been of great benefit to New Zealand already - and that's likely to continue.
-
Nitrogen fertiliser
Reducing the use of nitrogen fertiliser is a measurable way of reducing the amount of nitrogen available in the soil to be transformed into nitrous oxide.
-
Once-a-day milking
Making a deliberate decision to milk only once a day throughout lactation can, under the right circumstances, reduce emissions and maintain profitability.
-
Low-emission feeds
Some supplementary feeds reduce methane emissions per unit of feed intake, while others help reduce nitrous oxide emissions by decreasing the amount of nitrogen excreted onto pastures.
-
Potential actions
Some practices and technologies have been promoted as options to reduce emissions, but research is ongoing to get them into the national greenhouse gas inventory and/or fully demonstrate their efficacy on farm.
How does nitrous oxide contribute to climate change?
As well as being highly effective at trapping heat, nitrous oxide is a long-lived greenhouse gas, like carbon dioxide. So, from a climate change perspective, the warming caused by every new nitrous oxide emission today adds to the warming caused by past emissions. Each emission lasts in the atmosphere for over a century, and the warming it causes continues for several more centuries after it has disappeared.
Globally, nitrous oxide has increased about 16% since pre-industrial times.
According to the latest New Zealand Greenhouse Gas Inventory, in 2019, nitrous oxide accounted for around 10% of New Zealand's total greenhouse gas emissions. That compares with carbon dioxide at around 46% and methane at around 42%. Nitrous oxide accounts for around 20% of New Zealand's total agricultural greenhouse gas emissions.
New Zealand's emissions of nitrous oxide have risen by about 50% since 1990—mostly as a result of the expansion of the dairy sector and increased use of nitrogen-based fertilisers. By far the largest source of nitrous oxide emissions is livestock urine deposited onto soils.
Under the Climate Change Response (Zero Carbon) Amendment Act, the Government has set a target to reduce long-lived greenhouse gas emissions (carbon dioxide and nitrous oxide) to net zero (that is, emissions are matched by removals) by 2050.
Where does nitrous oxide come from?
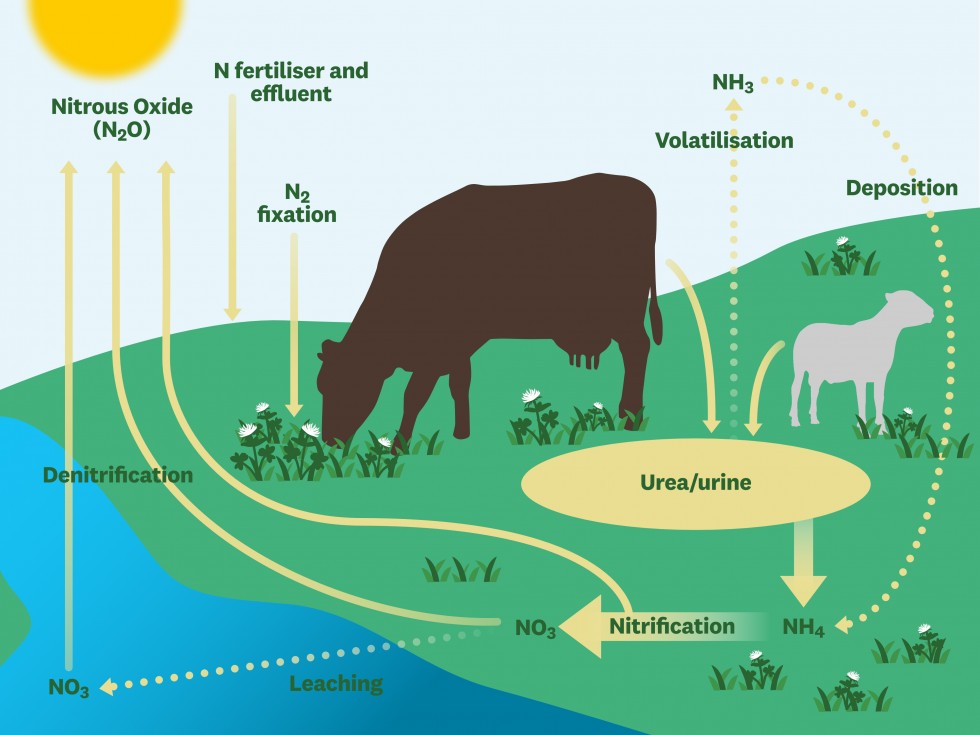
Nitrous oxide emissions come from a range of sources, including fossil fuel combustion. In agriculture, nitrous oxide is emitted into the atmosphere when micro-organisms act on nitrogen introduced to the soil via animal urine and dung, synthetic fertilisers and legumes. About 1% of nitrogen in the soil, from any source, is lost as nitrous oxide.
Many farmers use nitrogen-based fertilisers, legumes or animal manures to enrich their soil with nitrogen and help crops and pastures flourish. When grazing ruminant livestock eat nitrogen-rich pastures or crops, they use only a fraction of the nitrogen consumed to support the production of meat and milk. They excrete most of it in urine and dung, which creates very concentrated nitrogen patches in the soil. For comparison, a urine patch can contain the equivalent of up to 1000 kg N per hectare, while fertiliser application rates are typically 30-50 kg N per hectare (although there may be several applications per year).
Nitrogen in the soil undergoes a range of transformations by micro-organisms (see diagram below). Some of the nitrogen is given off as ammonia through a process called volatilisation. Over time, this is redeposited as nitrogen in rainfall and eventually gives rise to nitrous oxide emissions.
One group of micro-organisms, called nitrifiers, transform ammonium (NH4) into nitrate (NO3) and a small amount of nitrous oxide is produced during this process, which is known as ‘nitrification’. Another group of micro-organisms, called denitrifiers, in turn convert some of the nitrate into nitrous oxide (a process called ‘denitrification’) which is emitted directly into the atmosphere.
Finally, some of the nitrate produced during nitrification isn't taken up by plants or converted by denitrifiers into nitrous oxide and just sits in the soil as nitrate. This can leach or run off in irrigation or rainwater and result in nitrous oxide emissions from rivers and lakes.
What can farmers do to reduce nitrous oxide emissions?
Currently, there are no simple solutions to reduce nitrous oxide emissions that will work consistently on all farms.
Right now, reducing emissions essentially comes down to reducing the amount of nitrogen in the farming system, and minimising the proportion of the excess nitrogen in the soil that is transformed into nitrous oxide.
Actions that will help you to achieve this can be found at the top of this page.
Considerable research is under way in New Zealand and overseas to identify and verify new approaches to reducing on-farm emissions of nitrous oxide. While none of these options is fully proven yet, a number are showing promise. You can find information on these approaches on the Future actions page.
Will initiatives to reduce nitrate leaching also reduce nitrous oxide emissions?
Leaching arises when there’s excess nitrogen in the soil. Nitrous oxide arises from nitrogen transformations in the soil: the more nitrogen there is, the more nitrous oxide is produced. Therefore, efforts to reduce the amount of nitrogen in the soil will reduce both leaching and nitrous oxide emissions.
Scientists are looking closely at practices they know will help reduce nitrate leaching to see how they affect nitrous oxide emissions. For example, they’re investigating if farmers can use catch crops such as oats to reduce excess nitrate in the soil at the end of winter, and how they can best schedule irrigation and fertiliser applications to minimise the risk of nitrogen loss via nitrate leaching and nitrous oxide emissions.
Case studies
Other helpful resources
For more on the sources and science of nitrous oxide, see:
- The NZAGRC website